|
This next step is about identifying anything that will require you to take action immediately. Identify any immediate actions or corrections required Whatever the case may be, build your team appropriately for addressing the issues identified.ħ. For example, you might need engineering, marketing, manufacturing or regulatory as part of your team. Once you’ve clearly determined the sources, products and processes involved with the CAPA, this will help you to identify the resources you really need on the team to help oversee the CAPA. Your CAPA should be treated like a project. You want to do this one time and show that you’ve been as thorough and holistic as possible.Ħ. You need to ensure that you’ve covered everything because now you’re doing a CAPA investigation. This includes all of your products, processes, quality events and any other source contributing to the CAPA. Many companies have received warning letters or observations for failing to track CAPAs properly, especially if that failure to track meant CAPAs were left open indefinitely. 
An important part of managing a CAPA is that there should always be a clear trail and a way to monitor progress. When a CAPA is initiated, you need to assign it a tracking or identification number for documentation purposes to help you keep track it throughout the process. The request is formally initiated as a CAPA If accepted, then you move onto step #4.Ĥ. If it’s rejected, you should document the reason and rationale behind why it was rejected. This next step is where the CAPA request will be either accepted or rejected by the board or party in charge of reviewing it. It’s a good habit to be regularly reviewing any issues that go on. Many companies use the Management Review Board (MRB) or Quality Review Board (QRB) to review these requests as a team.Īn important tip here - it might be helpful to set up some sort of frequency for these meetings, not just for CAPA but for all quality events or issues and to keep a finger on the pulse. Who should be reviewing these? Usually, you’d have someone from quality, such as a quality manager, playing a critical part, but it doesn’t have to be a singular function involved. Your sources and resources need to determine whether the CAPA should be accepted and formalized into the system. The appropriate resources should review the request Be very clear about what the issue is and how it came to light.Ģ. For example, was the CAPA issue picked up at an audit, from a customer complaint or from a nonconformance? You need to corroborate the need or request that you are submitting. You need to identify your sources for this CAPA and record them. Create a CAPA request and submit for review 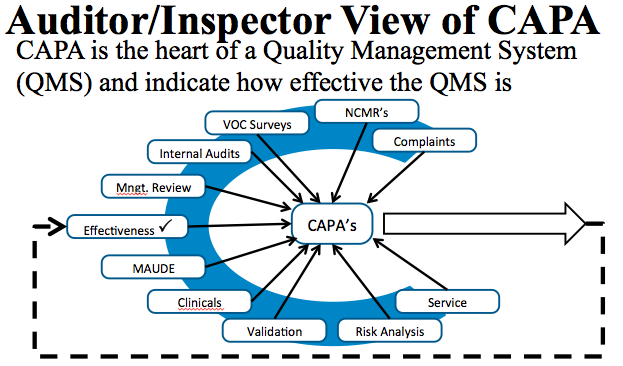
Once you have something that’s worthy of a CAPA, here are the steps:įree Download: Click here to download your own PDF copy of the CAPA Process Infographic.ġ. Remember, it should be reserved for those systemic issues - every issue is not a CAPA! You need to be diligent, but also apply scrutiny to what does and doesn’t require CAPA. 
The key thing about the CAPA process is that there are a lot of different processes that feed into it. This time, we’re looking at a step-by-step process that, if done well, should help any company to get a good result from their CAPA process. CAPA is an area that we talk about often, mainly because it’s still such a big issue for medical device companies.With CAPAs being a key issue indicated in FDA warning letters and observations, it’s important that companies have it all together and understand how to deal with them.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
